 Q: Why is a negative times a negative positive?.Q: Why was it so hard to take a picture of a black hole? What are we even looking at?.Q: Could we get rid of CO2 if we pumped it through a pipe into space?.Q: How hard is it to build a space elevator? What’s the point?.Q: What’s the point of going to the Moon?.Q: Do we actually live in a computer simulation?.Q: What is quantum supremacy? Is it awesome or worrisome?.Q: Why are the laws of quantum mechanics so strange? Does it mean that we’re missing something?.Q: Is silicon life possible? Why all the fuss over carbon-based life?.A Quantum Computation Course 2: Quantum Harder. 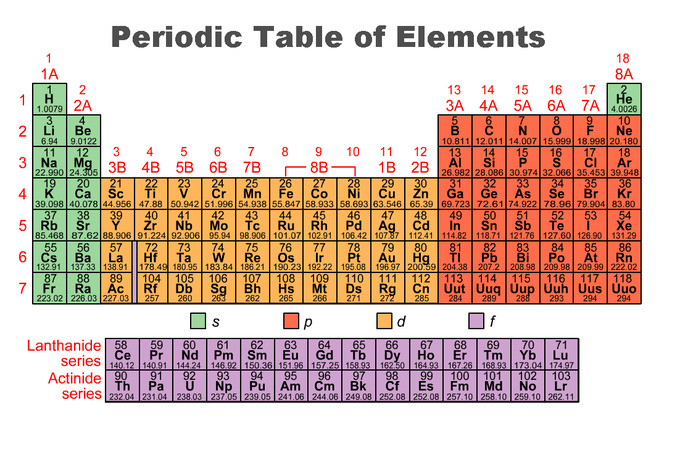
A Quantum Computation Course 3: Rise of the Quanta.A Quantum Computation Course 4: Full Measure.Q: What if the particles in the double slit experiment were conscious? Could you ask them which slit they went through afterwards?.Q: How big is the universe? What happens at the “edge”?.Q: How likely is it that there’s dark matter in me right now?.Q: Is getting plasma really hot the only way to initiate fusion?.Q: If the universe gets split in the Many Worlds Interpretation, then why aren’t all probabilities 50/50? Does stuff get spread thinner and thinner with each split?.Q: Is it possible to create an “almanac” of human behavior that predicts everything a person will do?.Q: How is matter created? Can we create new matter and would that be useful?.This would make Gd3+ need to be in the “g” orbital. One third of the electrons in the g-subshell are found in the “g” orbital. There are 6 electrons in the “g” orbital (one third of 6), so there are 3 electrons in the ” h ” orbital. The 6 electrons in the g-subshell are distributed amongst the 3 orbitals that make up the g-subshell, so Gd3+ would need to be in the “g” orbital. The reason Gd3+ doesn’t fit is because the g-subshell contains 6 electrons (there are 6 protons in the nucleus). The only way to get an electron to settle into a “g” sub-shell is to create element 121 or higher and then, before all of it radioactively decays into lighter elements (which has a way of ionizing everything nearby), you need to get it cooled off and electrically conductive. “So to get an electron to settle into a “g” sub-shell you’d need to create element 121 or higher and then, before all of it radioactively decays into lighter elements (which has a way of ionizing everything nearby), you need to get it cooled off and electrically neutral.” Because most of the energy is in the form of heat, fusion reactions are usually slowed down by the surrounding material which soaks up the heat.Ī phenomenologically important characteristic of fusion reactions is that they are relatively slow. The electrons that are stripped off the nucleus of the atom are released back into the surrounding area. 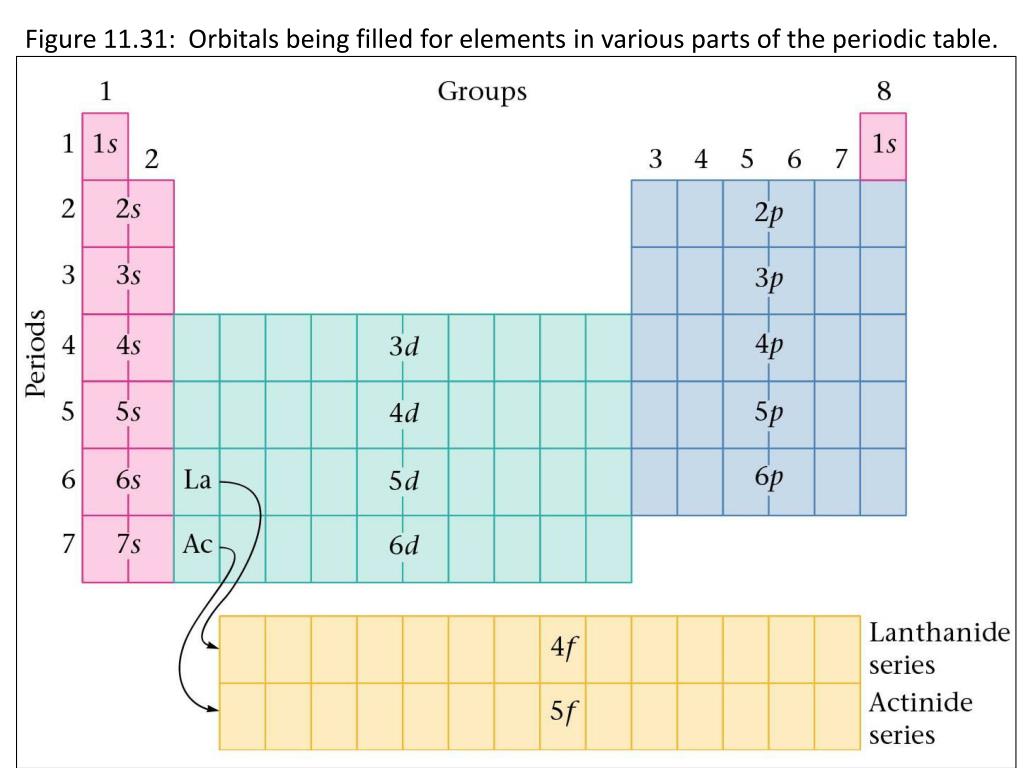
Then, before you can say “Nobel please”, it’s gone. So to get an electron to settle into a “g” sub-shell you’d need to create element 121 or higher and then, before all of it radioactively decays into lighter elements (which has a way of ionizing everything nearby), you need to get it cooled off and electrically neutral. In order to fill up all of their shells and sub-shells, atoms need to be in a relatively cool environment. Generally speaking, the processes used to create new elements are energetic enough (hot enough) that the atoms are formed in an ionized state. The half-life of element 118 is about 1/1000 of a second, and 121’s is probably shorter. The higher the atomic number, the less stable the element is. This is all a bit of a moot point however. For comparison, uranium is element number 92. 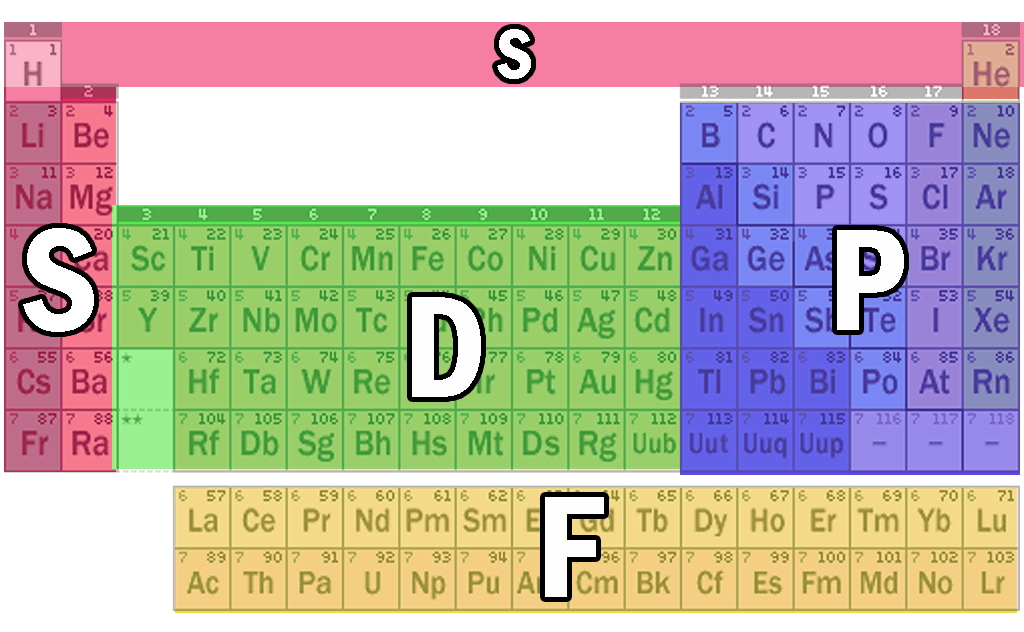
However, the largest found so far is 118 (so close!). The first atom that would need a “g” orbital would be element 121. The different "levels" of the periodic table are caused by the strange filling order of the electron orbitals. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
